
Quick answer: ISO 9001 does not require “lots of paperwork.” It requires certain documented information and records (Clause 7.5) that prove your Quality Management System (QMS) is planned, controlled, and working. What else you need depends on your processes, risks, industry, and how you deliver products or services.
Who this is for: Small businesses preparing for a first ISO 9001 audit, growing companies formalising their QMS, and multi-site organisations that need consistent documentation across teams and locations.
A practical note: The “mandatory documentation” list below ties to ISO 9001’s documented information controls (Clause 7.5) and is supported by evidence requirements such as internal audits and management reviews (commonly covered under Clauses 9.2 and 9.3).
Why Most Businesses Struggle with ISO 9001 Documentation (And What They Often Miss)
When businesses begin implementing ISO 9001, one of the biggest challenges they face is documentation.
Most organizations assume ISO 9001 is about creating a large number of documents. In reality, the problem is not the quantity, it’s the clarity and relevance of documentation.
Many companies either:
- Over-document (creating unnecessary paperwork), or
- Under-document (missing critical requirements)
Both approaches can lead to audit failures or inefficient systems.
In Practice:
A small service company created over 50 documents for ISO compliance, but failed the audit because employees were not following them.
Key Insight:
Organizations that focus on practical, process-driven documentation rather than volume see up to 30% better audit outcomes.
Understanding ISO 9001:2015 Documentation Requirements: What Is Actually Mandatory vs Optional
A common misconception is that ISO 9001 requires extensive mandatory documentation. The truth is, the standard only specifies certain documented information, while allowing flexibility in how it is maintained.
Mandatory vs Optional Documentation
| Mandatory (Required by Standard) | Optional (But Recommended) |
| Quality policy | Standard Operating Procedures (SOPs) |
| Quality objectives | Process flowcharts |
| Scope of the QMS | Work instructions |
| Records of competence and training | Risk registers |
| Monitoring and measurement records | |
| Internal audit records | |
| Management review records | |
| Non-conformity and corrective action records |
In Practice:
A company with fewer but well-structured documents often performs better in audits than one with excessive paperwork.
Key Insight:
Effective documentation is about control and usability, not volume.
What Auditors Expect Beyond the Minimum
Auditors don’t just check for required documents; they evaluate how effectively your system works.
What Auditors Look For
- Evidence of implementation
- Consistency in processes
- Employee awareness
- Traceability of records
Key Insight:
Even if you meet minimum requirements, poor implementation can lead to non-conformities.
The Complete ISO 9001:2015 Mandatory Documentation List You Cannot Ignore
This section outlines the essential documents every organization must maintain for compliance.
Quality Policy: Defining Your Organization’s Commitment to Quality
A formal statement that reflects your organization’s commitment to quality and continuous improvement.
Quality Objectives: Setting Measurable Goals That Drive Performance
Objectives must be:
- Measurable
- Aligned with business goals
- Regularly monitored
In Practice:
An organization set a goal to reduce customer complaints by 15% within one year.
- Scope of the Quality Management System (QMS)- Defines the boundaries and applicability of your QMS.
- Documented Processes and Controls- Ensures consistency across operations by defining how processes are performed.
- Records of Competence, Training, and Awareness- Demonstrates that employees are capable of performing their roles effectively.
- Monitoring and Measurement Records- Tracks performance metrics and process effectiveness.
- Internal Audit Program and Reports- Shows that your organization regularly evaluates its own system.
- Management Review Records- Confirms that top management is actively involved in reviewing performance.
- Non-Conformity and Corrective Action Records-Document issues and how they are resolved to prevent recurrence.
Key Insight:
These records are critical because they prove your system is implemented, not just documented.
Industry-Specific Documentation: How Requirements Differ for Manufacturing, IT, and Healthcare
ISO 9001 works across all industries, but the documents used in each business can be very different. Here’s how documentation usually differs by sector.
| Industry | Key Documentation | Valuable Practical Point | Middle East-Relevant Example |
|---|---|---|---|
| Manufacturing | Process control documents, inspection records, supplier evaluation records | Documentation helps standardize production, reduce defects, and improve traceability across batches and suppliers. | A manufacturing company can reduce defects by standardizing inspection checklists and supplier approval records across all production lines. |
| IT & Software | Project plans, change logs, client requirement documents | Clear documentation improves delivery consistency, version control, and change management. | A software company serving enterprise clients in the Gulf can use approved requirement documents, test records, and release logs to reduce rework and avoid delivery confusion. |
| Healthcare | Patient documentation, risk management records, compliance protocols | Healthcare documentation supports patient safety, traceability, and compliance. | A hospital can improve patient safety by standardizing treatment records, handover forms, and incident reporting procedures. |
| Education | Curriculum records, learner assessment records, faculty competence records, LMS access/change logs | In education, documentation supports consistency in learning delivery, governance, and student experience, especially where digital learning is involved. | A university or training institute in the Middle East using blended learning can standardize course approval records, attendance logs, assessment controls, and e-learning platform change records to improve consistency and governance. |
| Oil & Gas | Permit-to-work records, contractor competence records, equipment inspection logs, incident and corrective action records | In oil and gas, documentation is critical because safety, contractor control, and operational discipline directly affect business continuity and risk. | An oil and gas contractor can use permit records, toolbox talk logs, equipment inspection records, and contractor competency files to show controlled field operations and stronger audit readiness. Aramco publicly highlights strict safety protocols and a safety culture that includes contractors and suppliers. |
| Textile | Fabric specifications, incoming material inspection records, buyer requirement sheets, supplier approval records, batch traceability records | Textile businesses need documentation to control quality, color/size consistency, supplier performance, and export readiness. | A Dubai-based textile sourcing or export business can standardize fabric inspection reports, buyer-approved specifications, and packing records to reduce complaints and improve shipment accuracy. Dubai’s apparel and textile trade fairs position the UAE as a major regional sourcing hub. |
| Import & Export | Commercial invoices, certificate of origin, packing lists, bill of lading/airway bill, import permits, customs clearance records | For import-export businesses, documentation is not only about quality. It also affects customs clearance, shipment release, and compliance. | A trader importing goods into the UAE or re-exporting through Sharjah can avoid delays by maintaining complete invoice, certificate of origin, packing list, and customs documentation. UAE and Sharjah customs list these as core trade documents. |
Reference Context for the Industry Examples
- Education: Based on UNESCO’s guidance on digital learning and education transformation.
- Oil & Gas: Based on Aramco’s public framework on strict safety protocols and protection of employees, contractors, and suppliers.
- Textile / Trade: Based on Dubai’s role as a global trade and business hub, supported by ecosystems such as DMCC.
- Import & Export: Based on official UAE guidance on trade documents such as the commercial invoice, certificate of origin, packing list, import permit, and airway bill.
Common Documentation Mistakes That Can Delay Your ISO 9001 Certification
Common Mistakes
- Copy-paste documentation
- Outdated or unused documents
- Lack of version control
- Poor document accessibility
In Practice:
A company failed the Stage 2 audit because employees were unaware of documented procedures.
How to Create ISO 9001 Documents That Actually Work (Not Just for Audit)
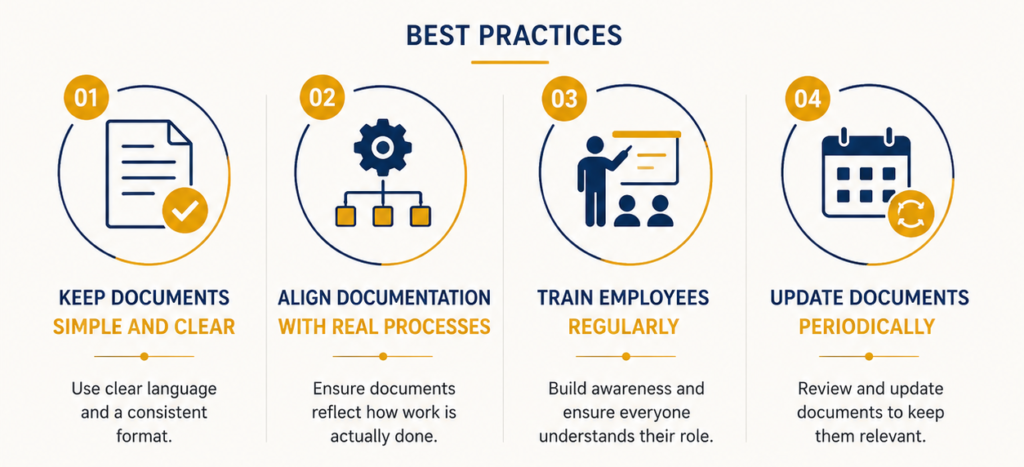
Real-World Example: How Businesses Simplify ISO 9001 Documentation Successfully
A mid-sized organization simplified its documentation by:
- Reducing redundant procedures
- Using flowcharts instead of lengthy text
- Digitizing records
Result
- Faster audits
- Better employee understanding
- Improved compliance
Preparing Your Documents for Audit: What Auditors Really Look For in 2026
As businesses evolve, audit expectations are becoming more practical and evidence-based.
Auditors Focus On
- Real implementation
- Data-driven decisions
- Risk-based thinking
- Continuous improvement
Key Insight:
Organizations using digital tools for documentation are better prepared for audits.
Is Your Documentation Ready? A Quick ISO 9001 Compliance Checklist
Ask yourself:
- Do you have all the mandatory documents?
- Are your records up to date?
- Are employees aware of procedures?
- Can you demonstrate process control?
If yes, you are on the right track.
Final Thoughts: Turning Documentation into a Business Advantage
ISO 9001 documentation should not be seen as a burden.
When done correctly, it helps organizations:
- Improve efficiency
- Reduce errors
- Ensure consistency
- Build customer trust
The goal is not to create documents, but to build a system that works.
Get Expert Support for ISO 9001 Documentation and Certification
Looking to implement ISO 9001 effectively in your organization?
Getting ISO 9001 documentation right is not just about passing an audit. It helps you build a system that is clear, practical, and easy for your team to follow. When your documents match your real process, your business becomes easier to manage, more consistent, and better prepared for certification.
If you want help with ISO 9001 documentation and certification, Guardian Middle East LLC is here to support you. We help businesses in Qatar and across the Middle East understand what documents are required, organize their Quality Management System, and prepare for audit with confidence.
Internationally Recognized Accreditation
Guardian Middle East LLC is based in Doha, Qatar. We support businesses with certification backed by internationally recognized accreditation.
Our certification support is recognized by:
- UAF (United Accreditation Foundation)
- IAS (International Accreditation Service, USA)
This gives your business stronger trust and wider acceptance in local and global markets.
Contact Our Experts
Mobile: +974 7770 2602 | +974 7213 7770
Email: info@guardian.qa
Website: www.guardian.qa
Start your ISO 9001 journey with expert guidance and build a system that truly adds value to your business.
Frequently Asked Questions
Do all businesses need the same ISO 9001 documents?
No. The required documents depend on your business type, processes, risks, and how you deliver products or services.
Can I pass an ISO 9001 audit with only the mandatory documents?
Not always. Auditors also check whether your system is working in practice, not just whether documents exist.
3. Are SOPs and work instructions mandatory under ISO 9001?
Not in every case. But they are often useful when processes need more control, clarity, or consistency.
4. What is the biggest documentation mistake companies make before certification?
Creating documents only for audit purposes without matching them to real daily operations.
5. How often should ISO 9001 documents be reviewed and updated?
They should be updated whenever processes change, gaps are found, or responsibilities are revised.
6. Can digital records be used instead of paper documents for ISO 9001?
Yes. Digital records are allowed as long as they are controlled, accessible, and properly maintained.
7. How do I know if my documentation is audit-ready?
If your documents are current, staff follow them, and you can show records as evidence, you are in a much better position for audit readiness.

Comments are closed